North Carolina woman describes what it was like to participate in COVID-19 Moderna vaccine trial

As the FDA prepares to review the the Moderna vaccine data next weeks, a North Carolina woman is describing what it was like to participate in the trial.
"I became unemployed, many of my colleagues became employed. And it became a domino effect throughout the entire economy. I was seeing more and more people becoming unemployed," said Gracie Howell, who had worked as a Sales Manager for private events and parties at a Durham hotel in March when the pandemic hit.
Two local Pfizer trial participants urge others to get COVID-19 vaccine when widely available
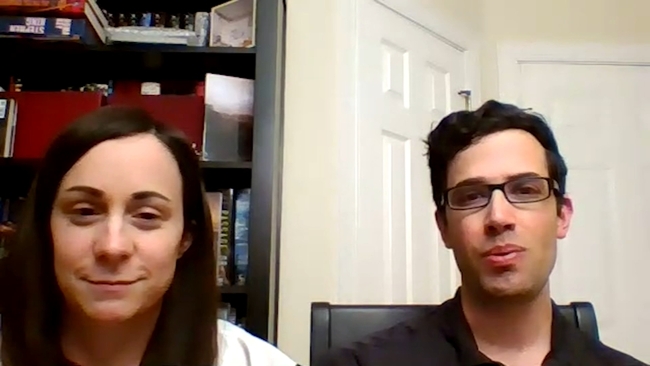
Howell ended up getting a job at Wegmans, a fortuitous landing spot in her hopes of participating in a vaccine trial.
"I was so thrilled when I found out that they were only taking people from essential businesses as clinical trial participants because they needed people who were being exposed the most to the public," Howell said.
UNC infectious disease specialist says Moderna vaccine trial participants were 'really dedicated'
She shared why she wanted to take part in the trial.
What will happen with NC's first 85,800 COVID-19 vaccine doses? State leaders explain
"Seeing the world was coming to a halt as COVID started to creep further and further into the population, just from studying history and pandemics, such as the Spanish Flu of 1918, it was pretty clear to me that for us in 2020, the solution was going to be a vaccine."
Howell cited her own studies in human biology and genetics from her time in college, as well as conversations with her dad.
"My father is a geneticist and he started his fellowship at NIH in 1960, so he's one of the most experienced living geneticists," Howell said.
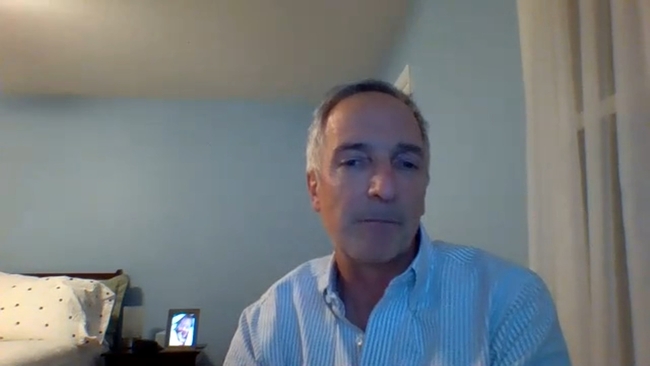
On the other side of that trial was Dr. Joseph Eron, the Chief of the Division of Infectious Diseases at UNC's School of Medicine, who serves as an investigator.
"It's a powerful experience. People stepping out and coming up and volunteering, and then knowing that it has such tremendous activity at least over such a short period of time that we could really make a difference in terms of limiting spread," said Dr. Eron.
He has spent the bulk of his career working on another epidemic.
"We were one of the first places in the country to do the first cocktail study for HIV," said Eron.
Like HIV, the COVID-19 pandemic has severely impacted underserved areas, stressing the need to include them in this research.
"About 2/3 of the people who were vaccinated at UNC were underrepresented minorities, either Black or Latinx. And those populations understandably have some trust issues, yet people came out," said Dr. Eron.
He highlighted the teamwork with Duke counterparts.
"Several Duke providers came over and got vaccinated on our trial, because we're not allowed to get vaccinated on our own research trial. So they came over and volunteered, (and) that was a big deal. We've had a lot of conversations together. People in Durham who need the vaccine go to Duke, and we deal with more people south. It's been very collaborative. I'm very excited for them and their work, and it couldn't be better. We're not the biggest metropolitan area in the country, but we have such great medical science at Duke and UNC," said Dr. Eron .
Both the Pfizer and Moderna vaccines were made using mRNA technology, a new way to deliver the vaccine to the body.
North Carolina's case count and hospitalizations have sharply increased over the past month, leading Governor Cooper to institute a modified Stay-At-Home order. The rise, which health officials believe will likely worsen in part due to recent Thanksgiving travel and gatherings, highlights the importance of the vaccine's impending arrival.
"I believe as more people receive the injections and receive the vaccines and see that their friends are doing well and not having significant problems, I think that will give people the reassurance they need to go ahead and get the vaccine," Howell said.
Howell does not know if she received the vaccine or a placebo, but said she felt no side effects from her injections.











